|
When the white light is responded to a medium, they get split consistent with their respective frequencies and wavelengths. Microwaves, actinic radiation, ultraviolet radiation, X-rays, and gamma-rays are kinds of radiation included within the Spectrum. Electromagnetic waves are further classified as portions, and ultraviolet rays and visual light are examples. Electromagnetic waves can travel in an exceeding vacuum at the speed of sunshine. A normal light features a large range of wavelengths with respective frequencies. We know that light is radiation composed of several frequencies of radiation. The Pauli exclusion principle asserts that no two half-integer spin particles (such as electrons) in an atom may occupy the same energy state simultaneously thus, the K-shell represents two different energy states, the L-shell eight, and so on. As each element encompasses a specific arrangement of electrons at discrete energy, it is appreciated that the radiation produced from such interactions is ‘characteristic’ of the element involved.Įlectron transitions from the L-shell to the K-shell, for example, yield x-ray photons with energies of 57.98 and 59.32 keV in a tungsten target. The electron within the K-shell is ejected (provided the incident electron’s energy is larger than the separation energy of the K-shell electron), abandoning a ‘hole’ with an emission of one x-ray photon, sometimes called a characteristic photon, with energy equal to the energy state difference between the outer and inner shell electron involved within the transition.Īs critical the continual spectrum of bremsstrahlung radiation, characteristic radiation is represented by a spectrum. This energy emission happens when a fast-moving electron collides with a K-shell electron. Characteristic Spectrum of radiation :Ĭharacteristic radiation may be a style of energy emission relevant for X-ray production. Emission spectra, absorption spectra, and continuous spectra are the three types of atomic spectra. The atomic spectra of atoms are a collection of all these wavelengths of the atom in a specific range of parameters, such as pressure, temperature, etc. When an electron is excited from one energy level to the next, it emits or absorbs light of a specific wavelength. Each chemical element’s atomic spectrum is distinct, and it is substantially responsible for matter’s colour.ĭuring transitions between different energy levels within an atom, an electron produces or absorbs a spectrum of electromagnetic radiation. The development of quantum mechanics hinged on the explanation of this phenomenon. The energy difference between the orbitals determines the frequency. An electron can hop from one fixed orbital to another: if the orbital it jumps to has higher energy, it must absorb a photon of a particular frequency if the orbital it jumps to has lower energy, it must emit a photon of a specific frequency. mostly all textbooks on the spectral lines of elements tend to only go up to uranium, however, textbooks go into much more detail for each element.An atom has a range of readily absorbed and emitted characteristic frequencies of electromagnetic radiation. You can see a list of of all the known spectral lines of elements on Wikipedia since it seems to have the most updated table. These heavier elements likely have their own spectral lines, however, due to all I stated above, it isn't exactly possible to measure. This isn't even considering how much this would cost. This would make measuring their spectral lines nearly impossible. It's incredible because some of the heavier and more unstable elements have insanely short half lives ranging from 100.5 days (the most stable isotope of fermium (Fm, 100)), to 0.69 microseconds (0.00069 milliseconds) (Oganesson (Og, 118)). I could not find any data as to why this may be, however, I believe it is simply because we could not test for the spectral lines of the heavier elements due to their instability and scarcity. Also, tables of the spectral lines of elements only seem to go up to the 99th element, Einsteinium (not including Astatine (At, 85) and Francium (Fr, 87). However, it is much more difficult to observe the spectral lines of liquids and solids due to how close together the atoms are. Each element has its own distinct spectral line and this fact can and has been used to identify an element. 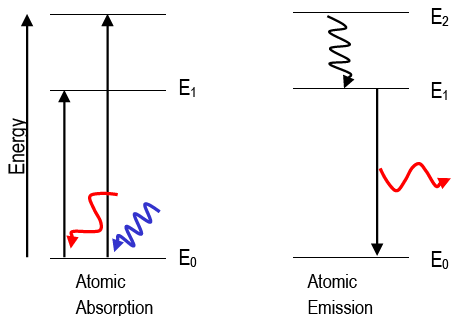 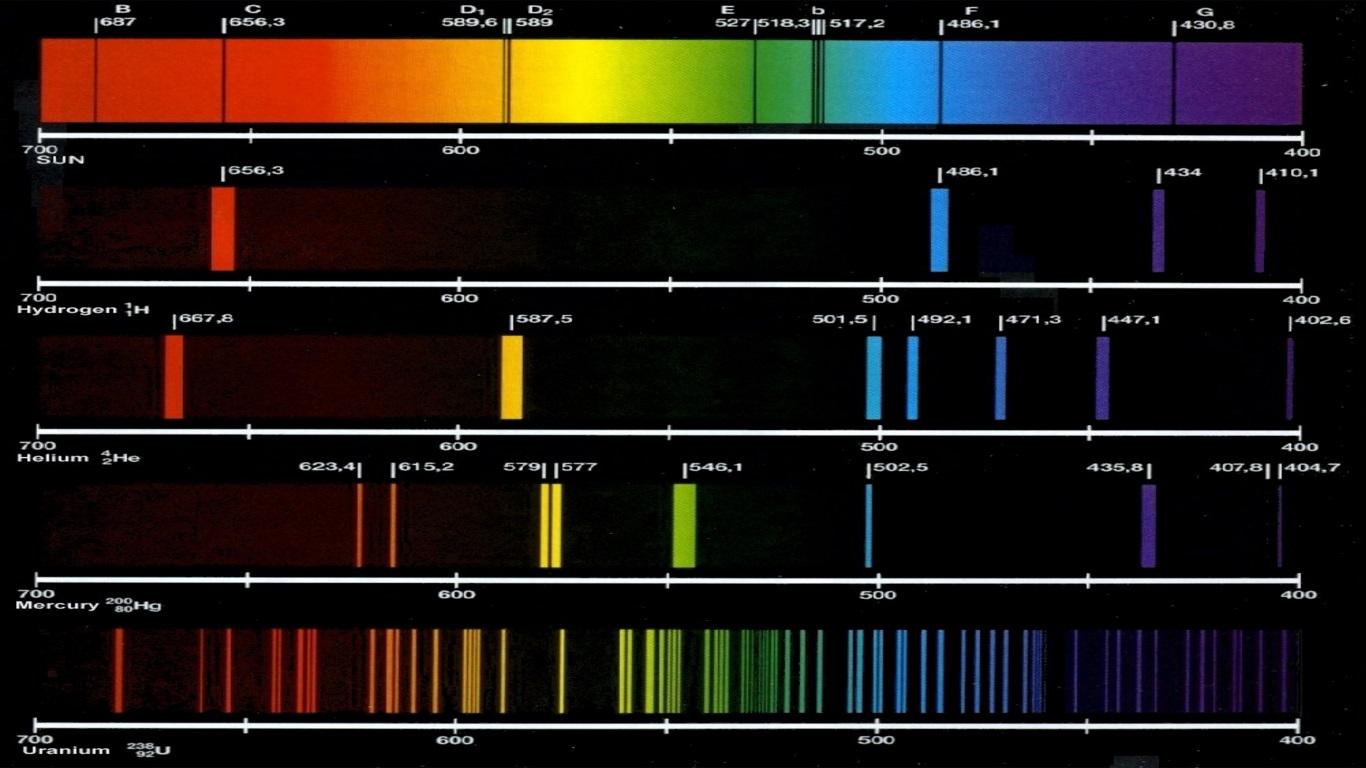
Actually, this phenomena can be observed with liquids and solids as well.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
